Abstract
Introduction: Loss of the Y-chromosome (LOY) is frequent in myelodysplastic syndromes (MDS) and observed as a single aberration in 3-4% of male MDS patients (pts). It is often clonal and not only age associated and confers a very good prognosis and a very low risk for leukemic transformation (Greenberg et al, Blood 2012; Schanz et al, JCO 2012). But LOY does not prove a hematologic disease per se (Arber et al, Blood 2016). To facilitate a better discrimination between age-related and clonal LOY, the aim of this study was to identify molecular mutations and cytomorphological features which might be characteristic for MDS with isolated LOY.
Methods: We included 291 pts in our analysis. The cohort comprised 199 pts with normal karyotype (NK) and morphologically proven MDS (excess blasts (EB) in 77/199 (38%) pts) and 92 pts with LOY. NK was defined by 20 normal metaphases or at least 10 normal metaphases and normal fluorescence in situ hybridization (FISH, Tab.1). Results from mutational analysis were available for all pts with NK and for 61 pts with LOY as single cytogenetic aberration in ≥3 metaphases. Seventeen core genes (Tab.2) were sequenced in all 260 pts by Sanger and/or next generation sequencing (NGS). In 134 pts further 28 genes (Tab.2) were analyzed using one of two NGS panels. In addition to these myeloid genes, the second NGS panel covered single nucleotide polymorphisms on the Y-chromosome which enabled determination of the LOY clone size. Detailed cytomorphology for the evaluation of dysplasia was performed by two experts (UG, UB) as previously described (Germing et al, Leuk Res 2012) in 41 pts, including pts with small LOY clones and with cytogenetic sub-clones.
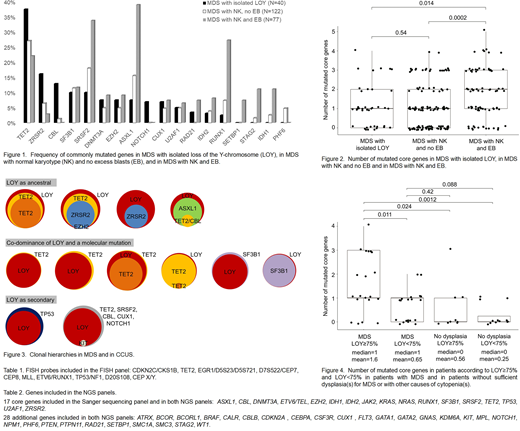
Results: Sequencing of 40 pts with LOY and morphologically proven dysplasia showed higher frequencies of mutations in TET2 (epigenetic regulator), ZRSR2 (splicing factor, located at Xp22.2), and CBL (kinase signaling) compared to MDS with NK (Fig.1). Amongst others, mutations in IDH1/2 (epigenetic regulators) and RUNX1 (transcription factor) were rare in MDS with LOY (Fig.1). The total number of mutated core genes did not significantly differ between MDS with LOY and MDS with NK and no EB (p=0.54), but it was significantly higher in MDS with NK and EB (p=0.014, Fig.2). To distinguish between LOY as ancestral or secondary mutation we sequenced 12 pts with MDS and cases we included as clonal cytopenia of undetermined significance (CCUS, pts with cytopenia(s) and molecular mutation and/or LOY≥75% of metaphases) (Wiktor et al, GCC 2000) using the second NGS panel that allowed determination of LOY clone size and detection of molecular mutations. Thereby, we identified four pts where LOY was most likely the founder aberration, two pts with LOY as secondary aberration in addition to ancestral molecular mutations, and six pts with co-dominance of LOY and a molecular mutation (Fig.3). Finally, we aimed to evaluate if the cut off of LOY≥75% (Wiktor et al, GCC 2000) can distinguish between age-related and clonal LOY in our cohort. In 41 pts analyzed in more detail, peripheral blood counts (hemoglobin: mean 10.4 vs. 9.7 g/dL; white blood count: 4.9 vs. 6.0x10(9)/L, platelets: 163 vs. 198x10(9)/L) and dysplasia of the individual cell lines (erythro-, granulo-, megakaryopoesis) did not differ significantly between LOY≥75% and <75%. Likewise, the VAF of molecular mutations did not significantly differ between MDS and pts without dysplasia according to LOY≥75% and <75%. Notably, for pts with LOY≥75% the number of mutated core genes was significantly higher in MDS compared to pts without dysplasia (p=0.024, Fig.4).
Conclusions: Cytopenia with isolated LOY seems to comprise a heterogeneous mixture of cytomorphologic and molecular subtypes. We identified pts where LOY definitely is part of the MDS clone that also harbors molecular mutations, indicating that LOY is not only age-associated in these cases. The low total number of mutated genes corresponds to the previously described favorable prognosis of isolated LOY (Schanz et al, JCO 2012; Greenberg et al, Blood 2012). Our data support the current guidelines that LOY does not prove a hematologic disease in the absence of diagnostic morphologic features. In these cases LOY might fulfill the requirements of a clonal mutation as described for clonal hematopoiesis of indeterminate potential (CHIP) or CCUS. In summary, our data suggest that MDS with LOY shows a characteristic molecular mutation profile.
Germing:Janssen: Honoraria; Celgene: Honoraria, Research Funding; Novartis: Honoraria, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.